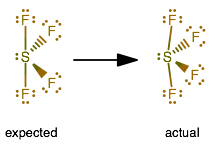
Let us study the VSEPR theory to predict the shape of iodine trichloride.Īs per VSEPR theory, the number of bonded (bond pair) and non-bonded (lone pair) valence shell electrons determine the shape and molecular geometry of the molecule. It is well understood by the valence shell electron pair repulsion (VSEPR) theory. We cannot predict the shape and molecular geometry of iodine trichloride from the Lewis structure. So, the Lewis structure of the iodine trichloride can also be represented as: The one shared electron pair represents the single bond. Therefore, these elements can be surrounded by more than eight electrons. It is one of the exceptions of the octet rule, i.e., the elements of the third period or beyond the third period of the periodic table have 3d electrons for bonding. We can observe that every chlorine atom is surrounded by eight electrons but a central atom, iodine, is surrounded by ten electrons. Hence, the Lewis structure of iodine trichloride would be: In the skeletal structure of ICl3, Iodine will be the central atom and all three chlorine atoms will surround it. The total number of valence electron are = 28 electrons. Hence, both of the atoms have seven valence electrons. The electronic configuration of I and Cl is 4d105s25p5 and 3s23p5, respectively. Completion of the lewis structure by fulfilling the criteria of the octet rule for every atom, if possible.Determination of the total number of valence electrons.The drawing of the Lewis structure of any compound consists of the following steps: It includes only electrons (bonding as well as nonbonding) that are present in the valence (outermost) shell of an atom.Īccording to the lewis theory, every atom prefers to have eight electrons around itself in the valence shell except Helium and Hydrogen. The 2D structure of any compound is best represented by drawing its Lewis structure. In the last, we will study its polarity, i.e., iodine trichloride is polar or nonpolar. We will approach chemical bonding by studying Lewis theory for knowing its Lewis structure and then we will discuss the hybridization of iodine atom in iodine trichloride molecule. Let us start to study the chemical bonding in iodine trichloride to understand its chemistry. The molecular weight of iodine trichloride is 233.26 g/mol. It is so reactive that it exists as a dimer, I2Cl6 in solid-state. ICl3 is bright yellow solid, which turns into red color upon exposure to light due to the formation of elemental iodine. They are more reactive than individual halogen atoms from which they are formed. These compounds are generally written as ABn where n= 1, 3, 5, and 7 A and B are less electronegative and more electronegative elements, respectively. Interhalogen compounds are molecules, which contain at least two different halogen atoms. ICl3, named Iodine Trichloride, is an Interhalogen compound.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
